
For centuries, batteries have been used to power devices and provide energy sources. From the early 1800s when Alessandro Volta invented the first battery to today’s modern lithium-ion cells, there has been a long history of evolution in battery technology.
The invention of the lead-acid cell by French physicist Gaston Planté in 1859 was a breakthrough that enabled the commercialization of electric vehicles for the first time.
Since then, significant technological developments have taken place such as nickel-cadmium and alkaline cells which paved the way for cordless tools and portable electronic gadgets like smartphones and laptops.
Today we are living in an era where renewable energies like solar or wind are also being used to charge our electronics without compromising on performance while reducing environmental impact at the same time.
It is amazing to think about how far we’ve come since those early days of battery technology!
The Emergence of Voltaic Piles & Primary Cells
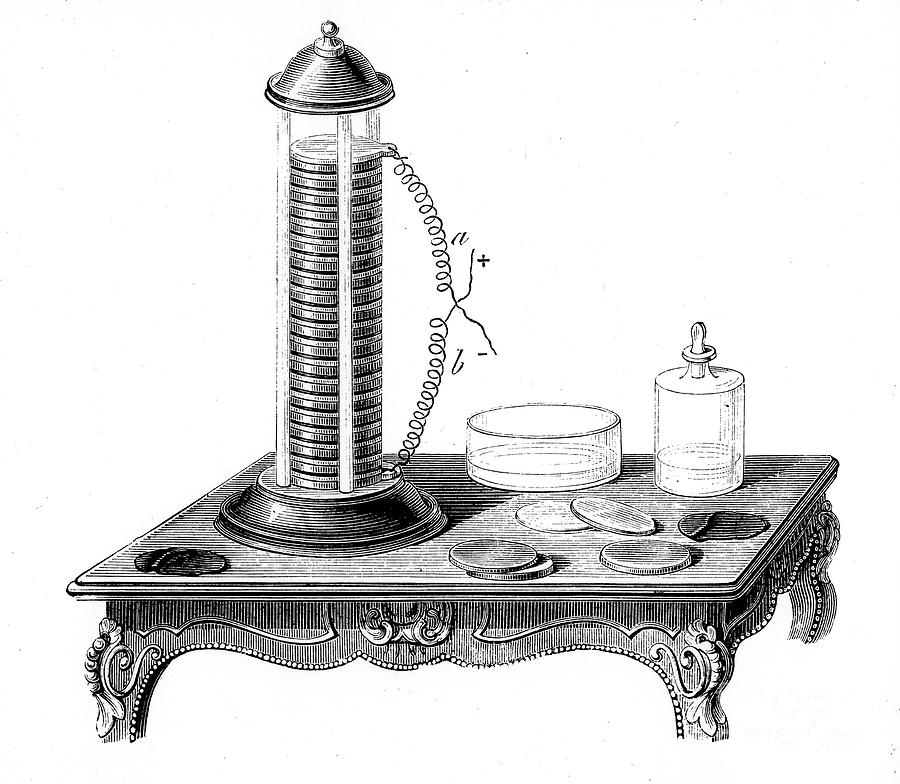
The invention of the voltaic pile in 1800 by Alessandro Volta marked a major milestone in battery technology. The voltaic pile was an early form of electrochemical cell and it was made up of alternating layers of zinc and copper along with brine-soaked paper or cloth as an electrolyte between them.
It provided a steady flow of electricity, which enabled scientists to conduct experiments that would eventually lead to the development of electric batteries. Primary cells were developed from the voltaic pile shortly afterward, allowing for greater control over electrical output.
These primary cells consisted of two different metals submerged into acid or alkaline electrolytes separated by porous material like ceramic cups or clay pots. This design allowed chemical reactions to take place at both electrodes, resulting in current being released when connections are established externally.
In addition to providing controllable power sources, these primary cells also paved the way for further research into rechargeable batteries and other forms of energy storage devices such as fuel cells and supercapacitors.
Secondary Cells: Rechargeable Batteries

Secondary cells, such as rechargeable batteries, have been a key component of the evolution of energy sources. These secondary cells can be charged and discharged multiple times without degrading their performance.
Rechargeable batteries offer numerous advantages compared to primary or non-rechargeable batteries because they provide greater capacity and a longer life span for powering electronic devices.
Furthermore, these types of cells can also be recycled which makes them more environmentally friendly than other battery types that use single-use chemicals that cannot be reused or recycled.
Secondary cells have become increasingly popular over time due to their convenience and usefulness in everyday applications ranging from electric vehicles to portable electronics like cell phones.
They play an important role in how we power our lives today with renewable energy sources like solar panels and wind turbines while reducing our reliance on fossil fuels like oil or coal for electricity generation.
Modern Day Lithium-Ion Batteries

Modern-day lithium-ion batteries are a formidable force in the world of energy storage. Developed in the early 1970s, these rechargeable cells are now used to power everything from cell phones and laptops to electric vehicles.
With their ability to store large amounts of energy for long periods, they have become an invaluable tool for modern life. Lithium-ion batteries also boast high-efficiency ratings and no memory effect when it comes to recharging – making them a great choice for anyone looking to move away from traditional fossil fuels and harness renewable energy sources instead.
With advancements in battery technology continuing at such a rapid rate, there is no doubt that lithium-ion batteries will continue to shape our future as we look toward sustainable solutions for powering our lives.
Conclusion

From the earliest days of civilization, batteries have been a key source of energy for humanity. From early experiments with static electricity to today’s ubiquitous lithium-ion cells, battery technology has seen incredible advances over time.
The ability to store power in large quantities and then discharge it on demand made possible many modern innovations such as mobile phones and electric vehicles.
As battery testing equipment continues to evolve and become more advanced, so too will our understanding of how best to use this incredibly versatile form of energy storage – a development that is sure to help shape the future for generations to come.








